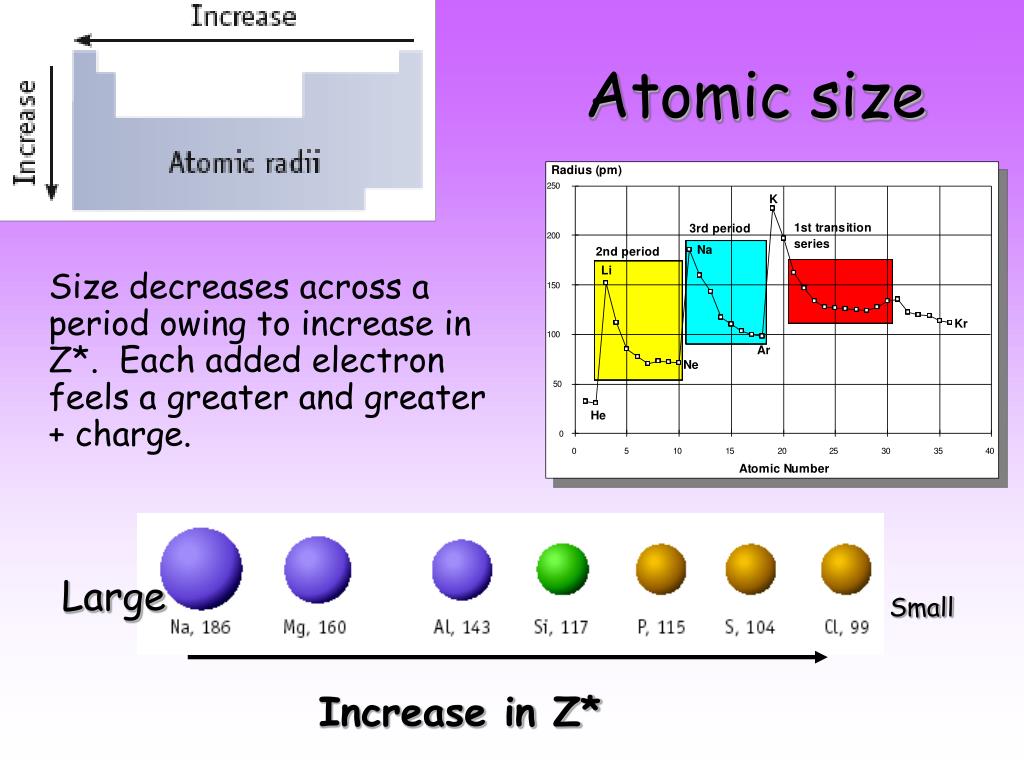 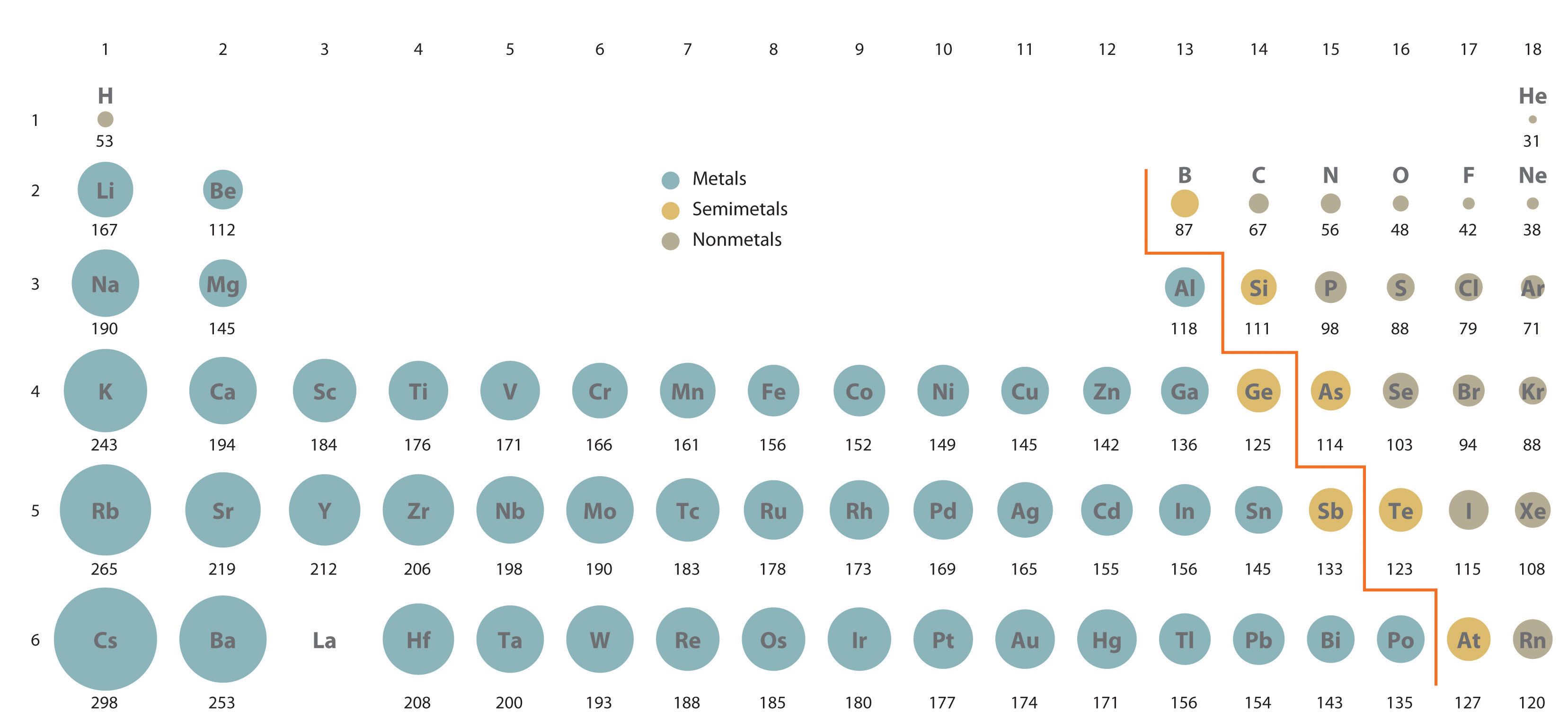
Image showing periodicity of the chemical elements for atomic radii (Clementi) in a 3D periodic table column style. Atomic Radius of the elements Hydrogen, 53 pm, Niobium, 198 pm, Thallium Helium, 31 pm, Molybdenum, 190 pm, Lead Lithium, 167 pm, Technetium, 183 pm, Bismuth. Read on to learn more about these two ways to measure an atom. The two are very similarand in some cases, even the samebut there are minor and important differences between them. Image showing periodicity of the chemical elements for atomic radii (Clementi) in a 3D spiral periodic table column style. Two measures used to describe atomic size are atomic radius and ionic radius. Image showing periodicity of the chemical elements for atomic radii (Clementi) in a spiral periodic table heatscape style. This dependence is now quantified for any atom within our definition of these properties. Two factors are important (1) the shell in which the valence electrons are found and (2) the strength of the interaction between the nucleus and the valence electrons. Image showing periodicity of the chemical elements for atomic radii (Clementi) in a periodic table heatscape style. 1 implies a strong dependence of electronegativity on atomic radius when individual atoms are compressed their size diminishes along with their electronegativity. Big Chemical Encyclopedia Atomic sizes The size of the atom is determined largely by its valence electrons because they occupy the outermost orbitals. Image showing periodicity of the chemical elements for atomic radii (Clementi) in a periodic table cityscape style. There is a correlation between the atomic radii as determined from these calculations and the radii of maximum charge density in the outermost shell of the atom. Atomic size is difficult to measure because it has. 
You should consult reference 1 for full details, but it is not light reading for most people. Atomic size is the distance from the nucleus to the valence shell where the valence electrons are located. These values are calculated using self-consistent-field functions (reference 1). The atomic radius of a chemical element is a measure of the size of its atoms. The values given here for atomic radius are calculated values using methods outlined in reference 1. (Heraldkeepers) - The report consists of the Atomic Layer Deposition (ALD) Market predominant achievements, R&D, new launches, responses, and nearby growth of the market’s maximum. Since the boundary is not a well-defined physical entity, there are various non-equivalent definitions of atomic radius. Sometimes in text books and other sources, the rather vague term "atomic radius" is not defined and in such cases it is therefore not clear what the values actually mean. The atomic radius of a chemical element is a measure of the size of its atom, usually the mean or typical distance from the center of the nucleus to the outermost isolated electron. Many references give table of atomic radii.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
